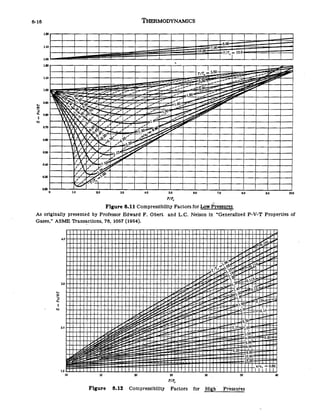
Compressibility factor (Z) for a van der Waals real gas at critical point is

Share your videos with friends, family and the world

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

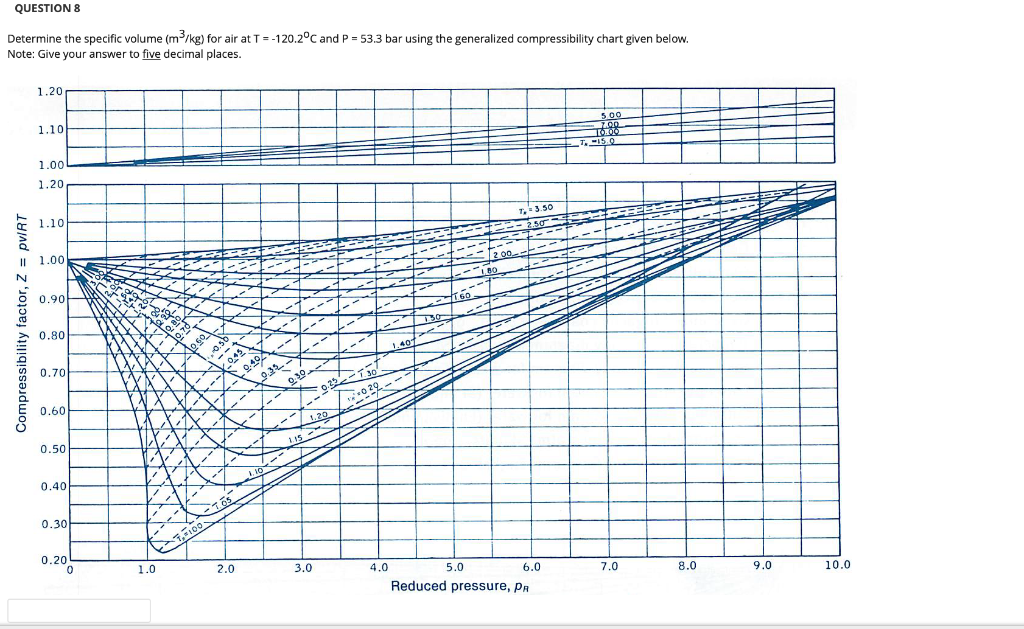
Solved Problem 2. ( 30 points) The ideal gas law can

Solved Problem 1. 15 points Find the compressibility factor

Physical Chemistry The Compression Factor (Z) [w/1 example]
Why there is different between the value of compressibility factor at critical point between real and ideal gas? - Quora

Calculate the critical temperature of a Van der Waals gas for which p(

Van der Waals equation - Wikipedia
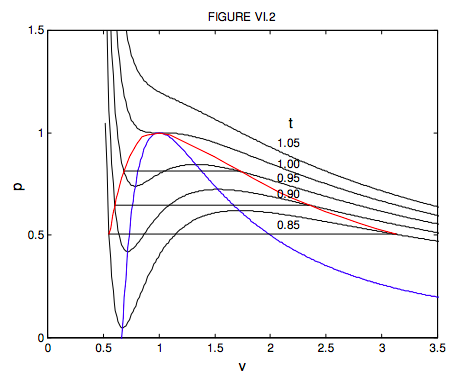
6.3: Van der Waals and Other Gases - Physics LibreTexts
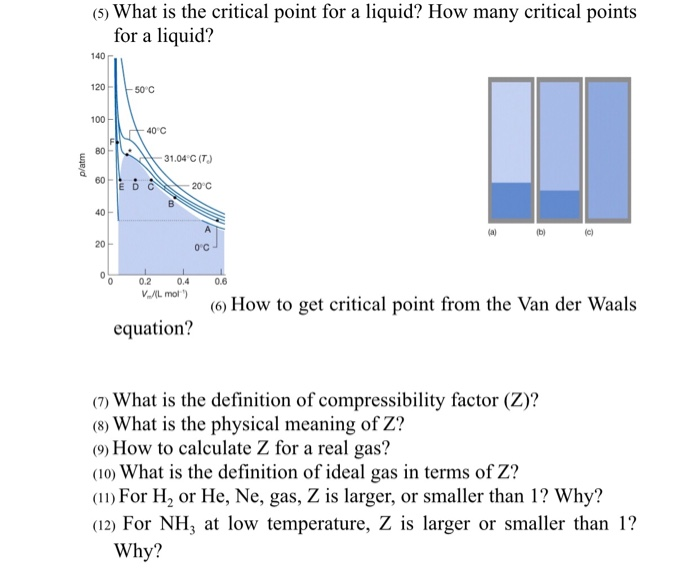
Solved (5) What is the critical point for a liquid? How many
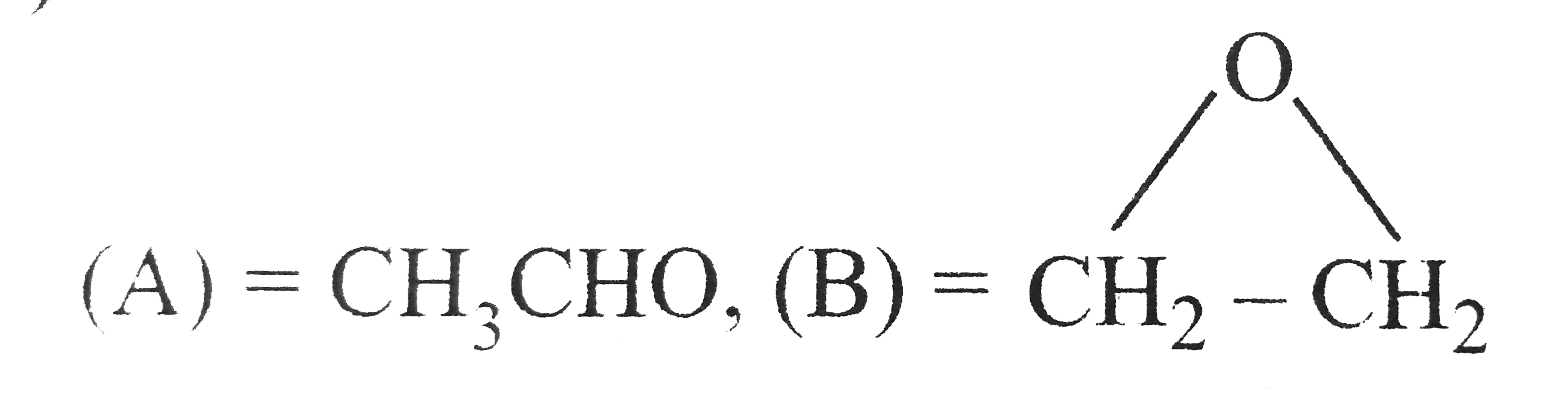
Complete Solutions to Mock Test 1 of chapter MOCK TEST of Class 11 book with complete answers and questions

Compressibility factor (Z) for a van der Waals real gas at critical po