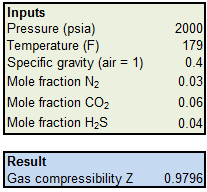
Answered: The virial equation of state gives the…

Description
Solution for The virial equation of state gives the compressibility factor Z of a gas as a function of the molar volume Vm or of the pressure P. PVm Z= B C =…
Computing the second virial coefficient

SOLVED: a) Virial equation of state of real gases is expressed as Z (PV / nRT) = 1 + (nB / V) + (n2C / V2). Derive the second virial coefficient B
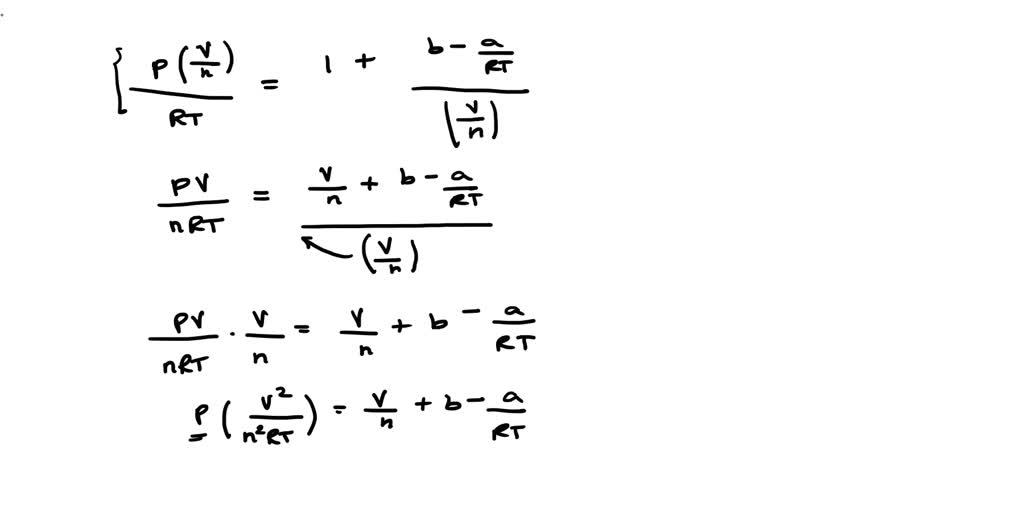
SOLVED: The virial equation of state of a gas describes deviations
Equation of state - Wikipedia

The virial equation for 1mole of a real gas is written as : PV=RT
A Viral Math Problem With 2 Answers Is Dividing the Internet

Equation of state - Wikipedia

Solved) - The mass density of water vapour at 327.6 atm and 776.4
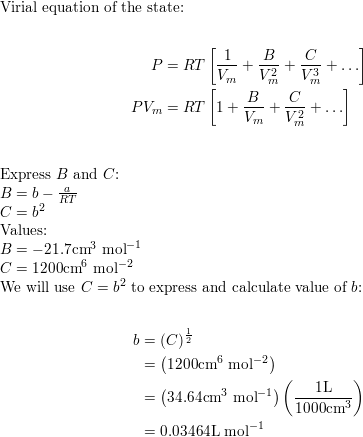
Express the van der Waals equation of state as a virial expa
Related products
You may also like

Swimwear – Miami HEAT Store

Designer Summer 2 Pcs Women Suit Set Vest+Pants Irregular Hem Wedding Tuxedo Formal Office Lady Wear Tailored Made Prom Dress - AliExpress

Auden Bra The Radiant Plunge Push-Up Lace Front Closure Lace Overlay Green 32AA

unas gafas de realidad aumentada de código abierto utilizan IA
$ 20.00USD
Score 4.9(416)
In stock
Continue to book
You may also like

Swimwear – Miami HEAT Store

Designer Summer 2 Pcs Women Suit Set Vest+Pants Irregular Hem Wedding Tuxedo Formal Office Lady Wear Tailored Made Prom Dress - AliExpress

Auden Bra The Radiant Plunge Push-Up Lace Front Closure Lace Overlay Green 32AA

unas gafas de realidad aumentada de código abierto utilizan IA
$ 20.00USD
Score 4.9(416)
In stock
Continue to book
©2018-2024, followfire.info, Inc. or its affiliates