Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H H is 0.64 and the bind length of F2 is 1.2. Electronegativities of H and F respectively are 2.1 and 4.1.What is the bond length of HF? 1)0.64 2)0.92 3)0.82 4)0.62
Bond length of H-H is 0-64 and the bind length of F2 is 1-2- Electronegativities of H and F respectively are 2-1 and 4-1-What is the bond length of HF- 1-0-64 2-0-92 3-0-82 4-0-62

Solvent induced mononuclear and dinuclear mixed ligand Cu( ii ) complex: structural diversity, supramolecular packing polymorphism and molecular docki - New Journal of Chemistry (RSC Publishing) DOI:10.1039/D0NJ03567J

Oxygen reduction electrochemistry at F doped carbons: A review on the effect of highly polarized C-F bonding in catalysis and stability of fuel cell catalysts - ScienceDirect

The X-X bond length is 1.5 Å and Y-Y bond length is 1.48 Å.If electronegativity values of X and

WO2015057963A1 - Fgfr4 inhibitors - Google Patents
Correct order of Bond Angle is 1)F2O>H2O>O3 2)O3>H2O >F2O 3)H2O

Write the increasing order of bond energies of H2,F2 and HF molecules.

Patai S., Rappoport Z. (Eds.) - The Chemistry of Organic Selenium and Tellurium Compounds. v.1, PDF, Functional Group
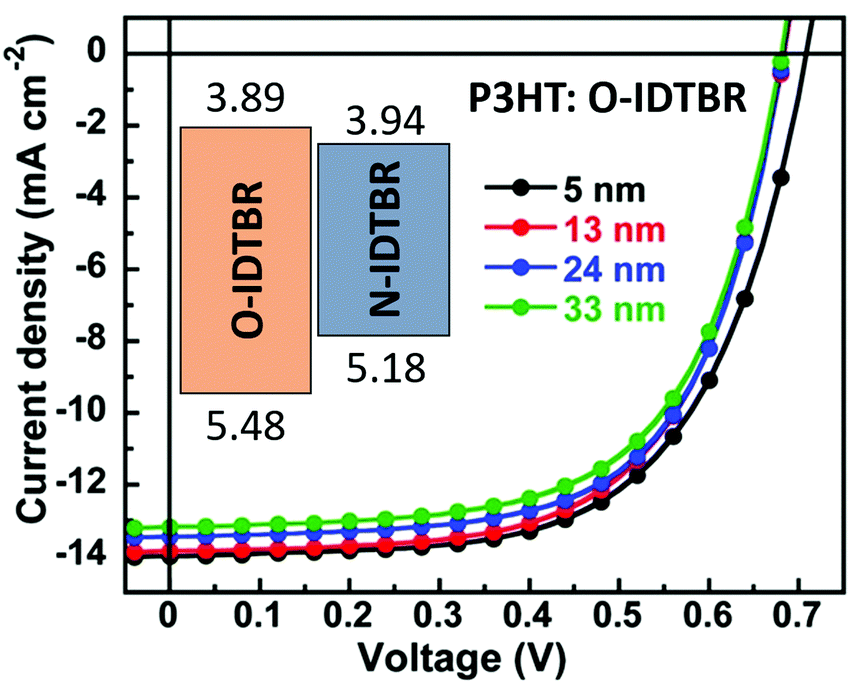
Interlayers for non-fullerene based polymer solar cells: distinctive features and challenges - Energy & Environmental Science (RSC Publishing) DOI:10.1039/D0EE02503H
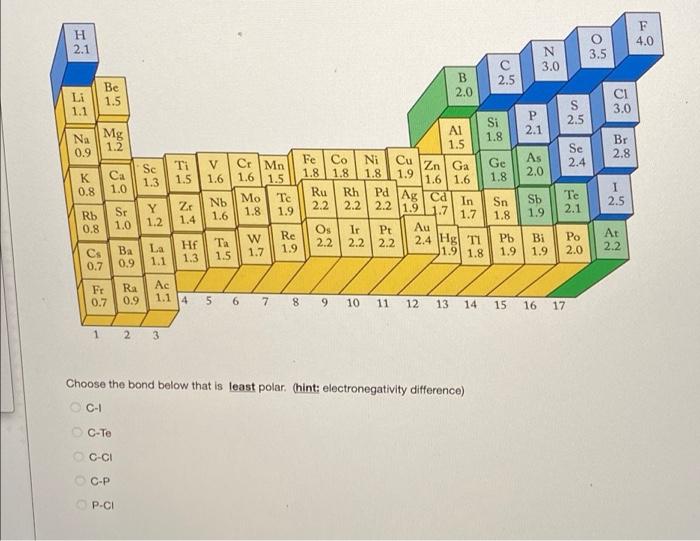
Solved Place the following in order of decreasing bond

PDF) From lithium phosphanide to a series of 2-amino-1λ3σ2-phosphaalkenes and some N-lithium phosphaguanidinates - syntheses, 2D NMR spectroscopic analyses and X-ray structure determinations
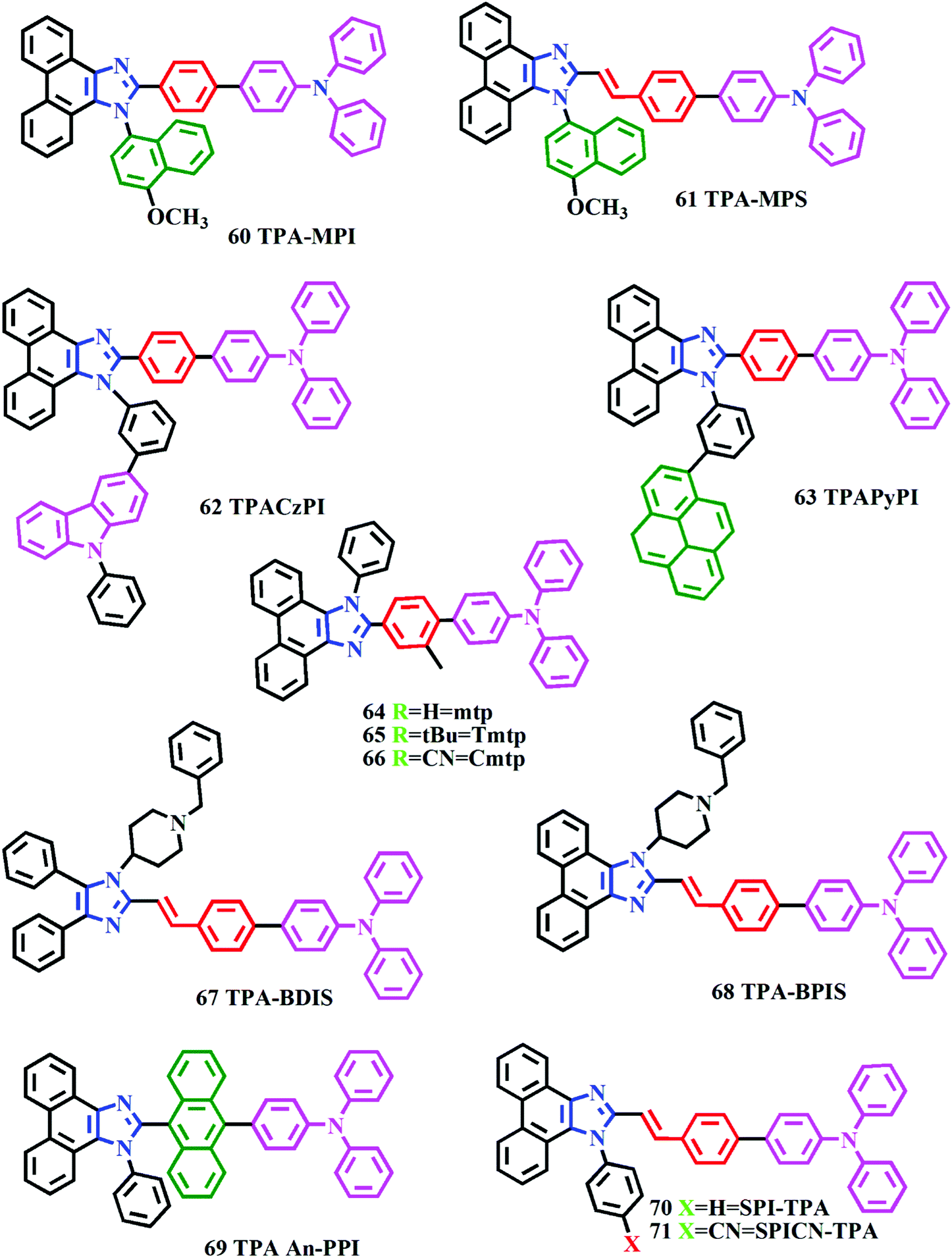
Recent development of phenanthroimidazole-based fluorophores for blue organic light-emitting diodes (OLEDs): an overview - Journal of Materials Chemistry C (RSC Publishing) DOI:10.1039/C8TC03689F
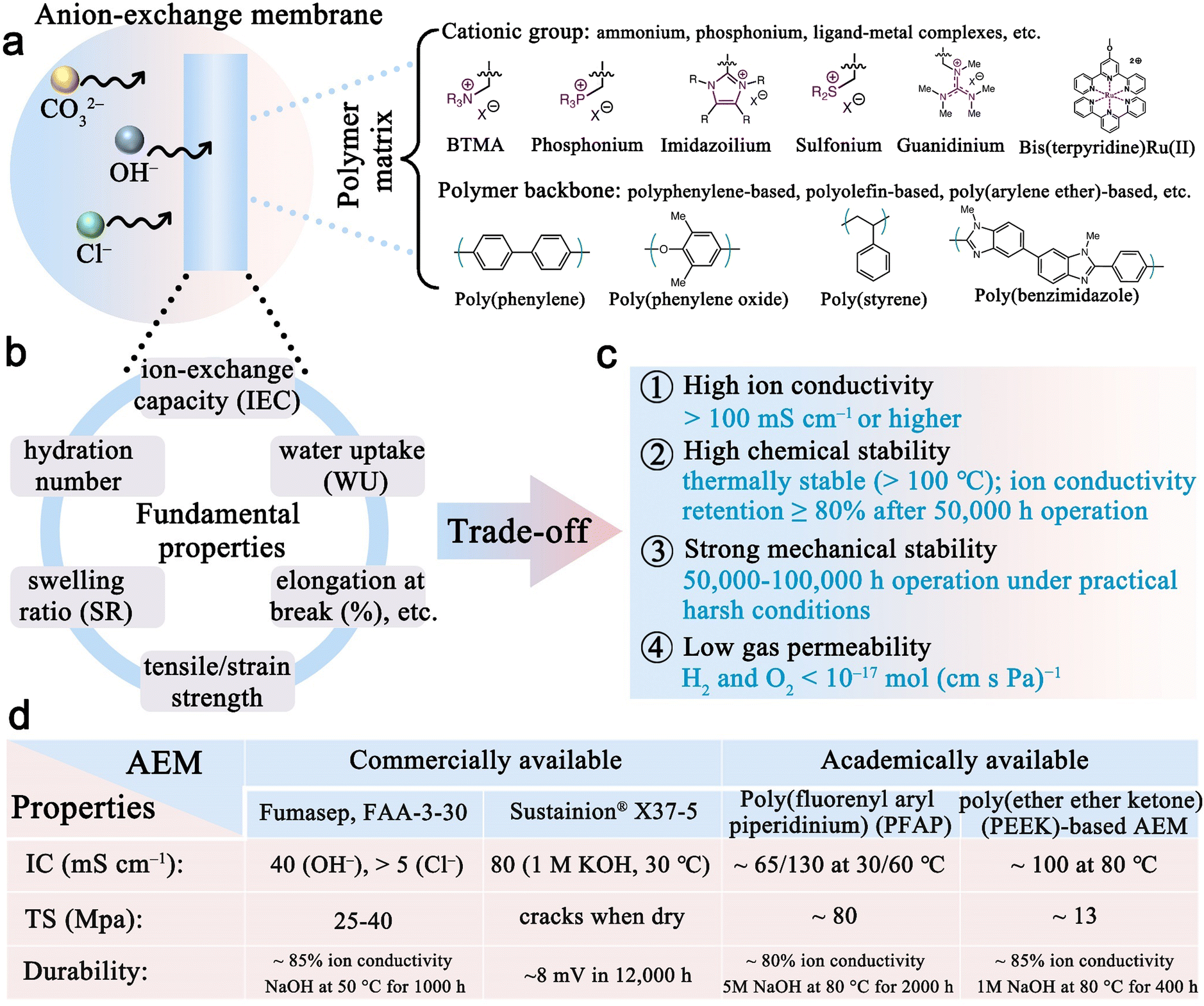
Anion-exchange membrane water electrolyzers and fuel cells - Chemical Society Reviews (RSC Publishing) DOI:10.1039/D2CS00038E

Solved The electronegativity is 2.1 for H and 4.0 for F.











