Telugu] What is compressiblity factor?

Description
It is the ratio of the actual molar volume of a gas to the volume of a perfect gas under the same conditions. For a perfect gas, the value of compression factor (Z) is 1.
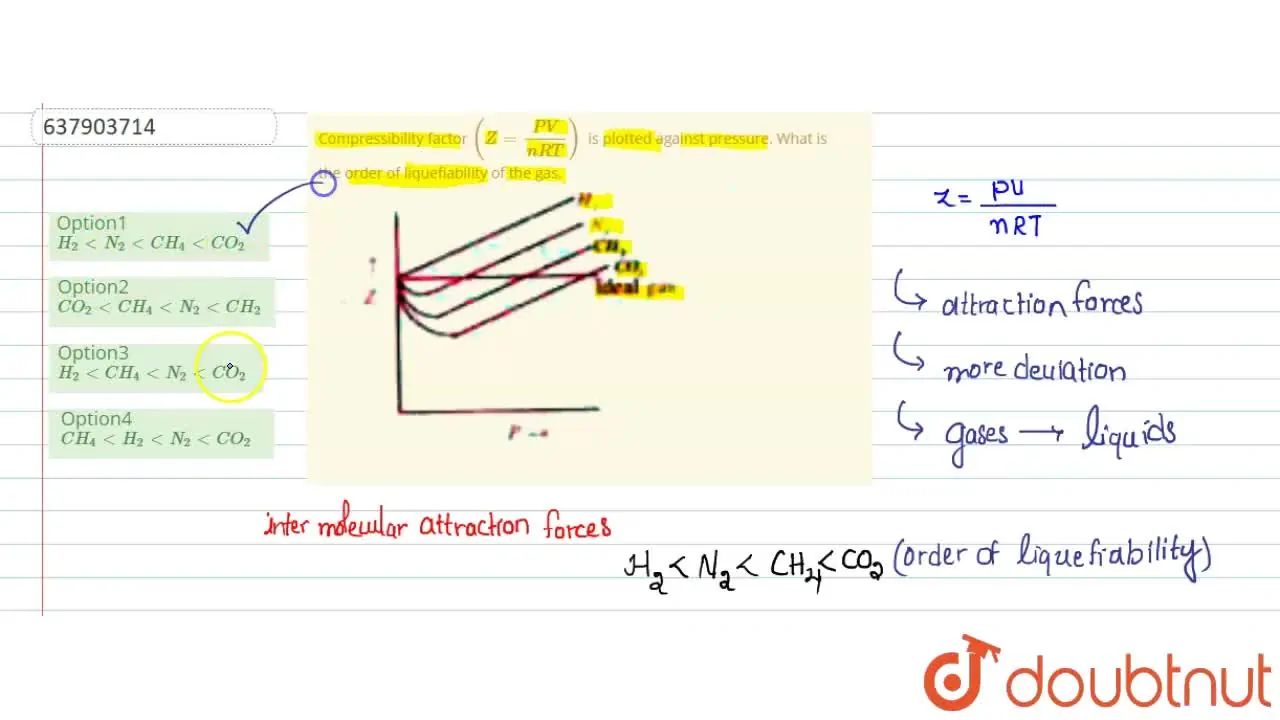
Telugu] Compressibility factor (Z = (PV)/(nRT)) is plotted against p

Telugu] Define compression factor. What is its value for a perfect-ga

Telugu] Compressibility factor (Z = (PV)/(nRT)) is plotted against p

Advanced Chemical Engineering Thermodynamics-31-July-2016

IIT JEE - JEE Mains-2014: Compressibility Factor- Van der Waals

Ideal Gas Equation - Heat and Thermodynamics Explanation In Telugu
Telugu] Using the compression factor explain the behaviour of real ga

Compressibility factor - Wikipedia

Compressibility Factor (Z)
Related products

At 273 K measurements on argon gave B = -21.7 cm$^3$ mol$^{

Pick only the incorrect statement.for gas A, a=0,the compressibility factor is linearly dependent on pressure.for gas C,aneq 0,bneq 0,it can be used to calculate a and b by giving lowest P value.for
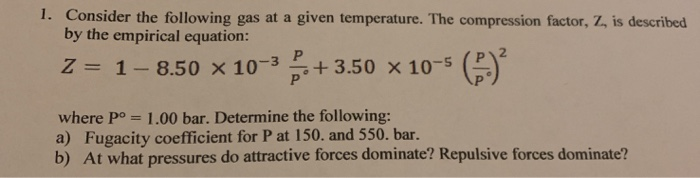
Solved 1. Consider the following gas at a given temperature.

53 pts!! The function f(x)= 7^x+1 is transformed to function g through a horizontal compression by a factor
You may also like

Nike Swoosh Front Zip Women's Medium-Support Padded Sports Bra. Nike IN

Solid Color Compression Wirefree Bras High Support Bra For Women Sports Seamless Underwear Lingeries Brasieres Para Mujer - AliExpress

LIBERTÉ Naio Leggings m. strukturmøster - Black Dot - KØB ONLINE

Kmart/Sears RN 42000 White Underwire Padded Push-Up Bra Size 38 C (28)
$ 25.99USD
Score 4.6(108)
In stock
Continue to book
You may also like

Nike Swoosh Front Zip Women's Medium-Support Padded Sports Bra. Nike IN

Solid Color Compression Wirefree Bras High Support Bra For Women Sports Seamless Underwear Lingeries Brasieres Para Mujer - AliExpress

LIBERTÉ Naio Leggings m. strukturmøster - Black Dot - KØB ONLINE

Kmart/Sears RN 42000 White Underwire Padded Push-Up Bra Size 38 C (28)
$ 25.99USD
Score 4.6(108)
In stock
Continue to book
©2018-2024, followfire.info, Inc. or its affiliates