Draft Guidance Document: Applications for Medical Device Investigational Testing Authorizations

This draft guidance document reflects Health Canada’s current thinking on Investigational Testing Authorizations (ITA) for medical devices and may be subject to changes as policy develops. The document clarifies application requirements and processes, including pre-ITA meetings, format for an ITA application and filing requests for revisions to an ITA.
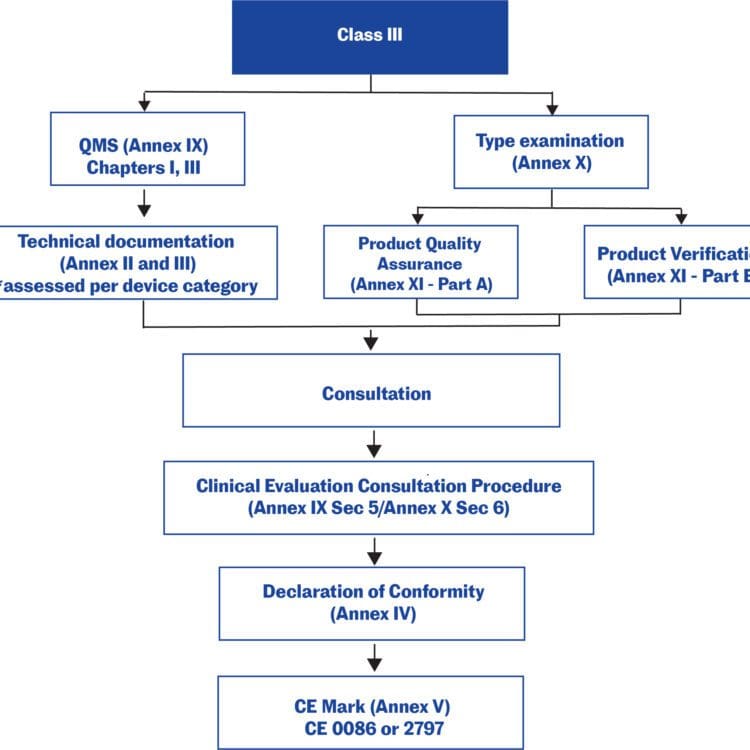
Medical device submissions: Placing a medical device on the market

Medical device submissions: Placing a medical device on the market
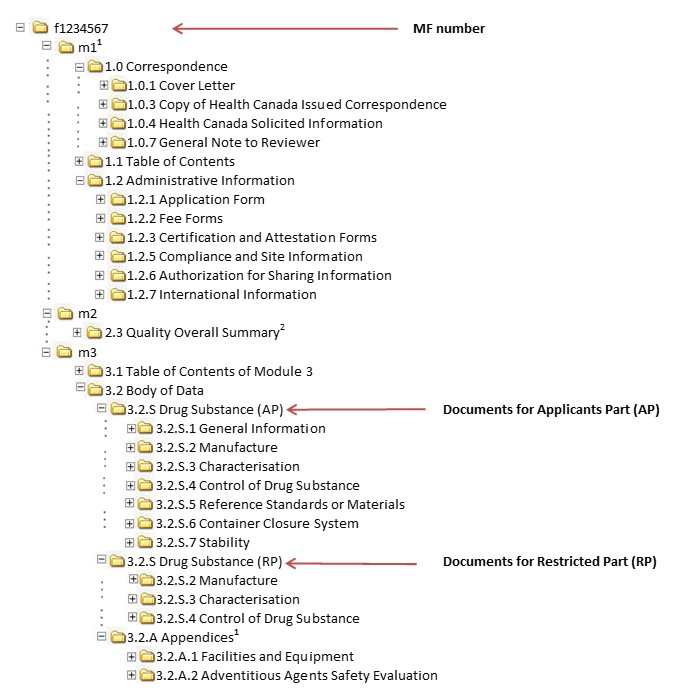
Guidance document: preparation of regulatory activities in non-eCTD format

Guidance Document - Creation of the Canadian Module 1 Backbone
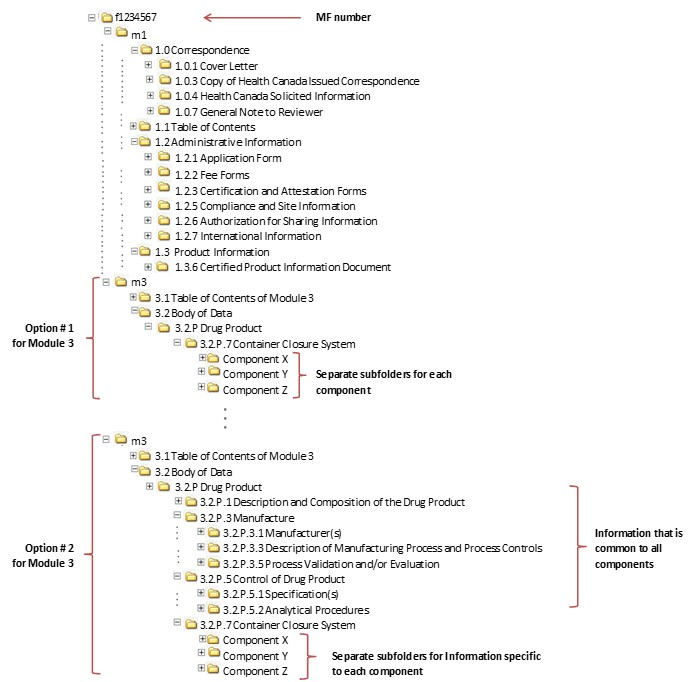
Guidance document: preparation of regulatory activities in non-eCTD format

Guidance document: preparation of regulatory activities in non-eCTD format

Canada's Health Canada - Global Regulatory Partners, Inc.
How FDA Regulates Artificial Intelligence in Medical Products

Canada's Health Canada - Global Regulatory Partners, Inc.

Steps involved in US FDA's Medical devices Validation Protocol Process
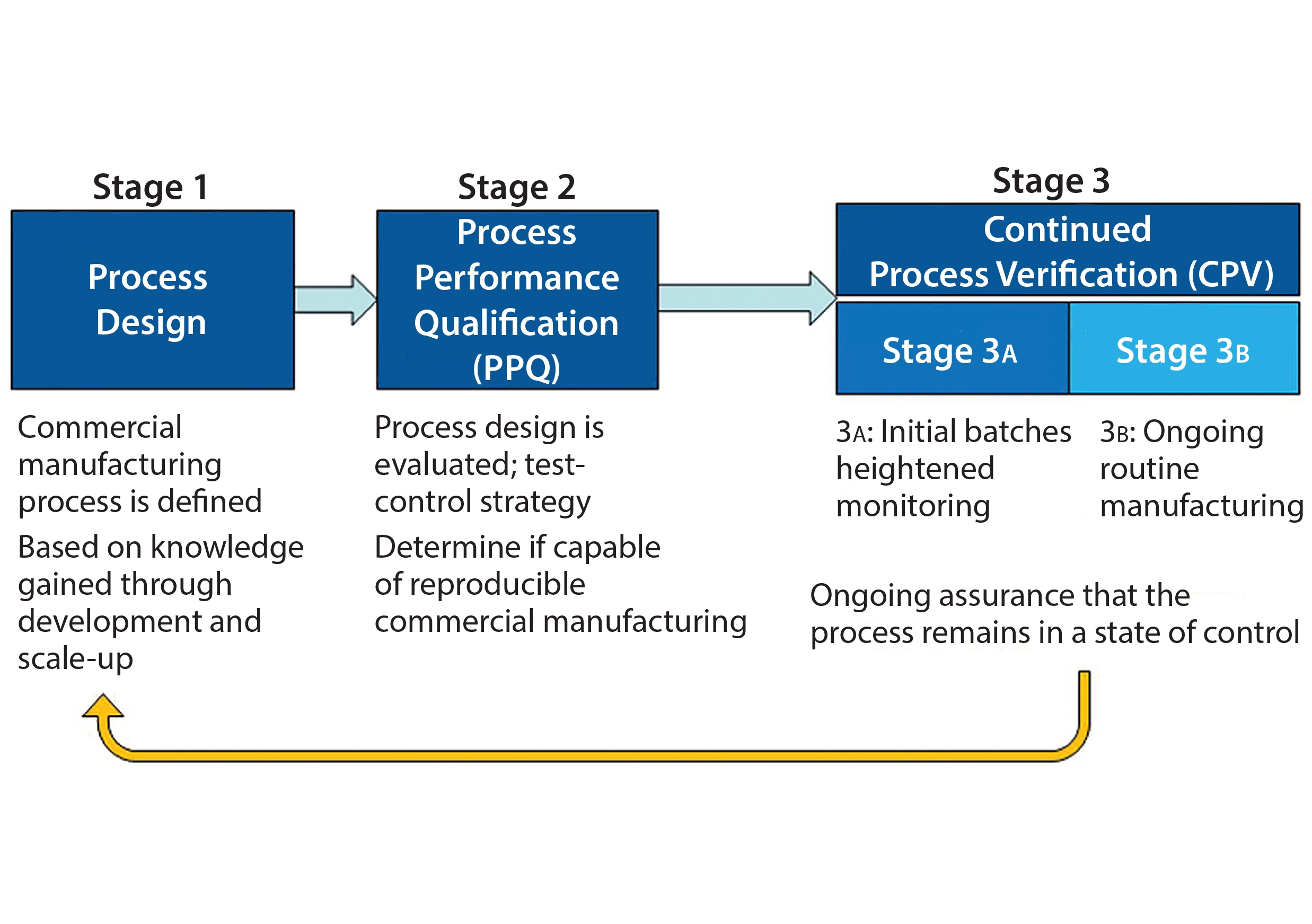
Continued Process Verification: Evolution of Biopharmaceutical Control Strategy - CMC Forum