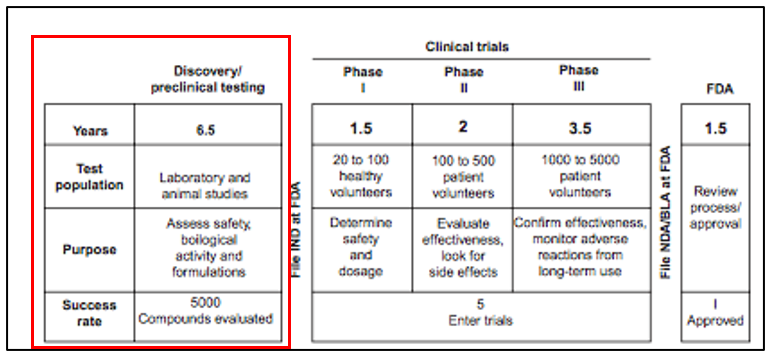
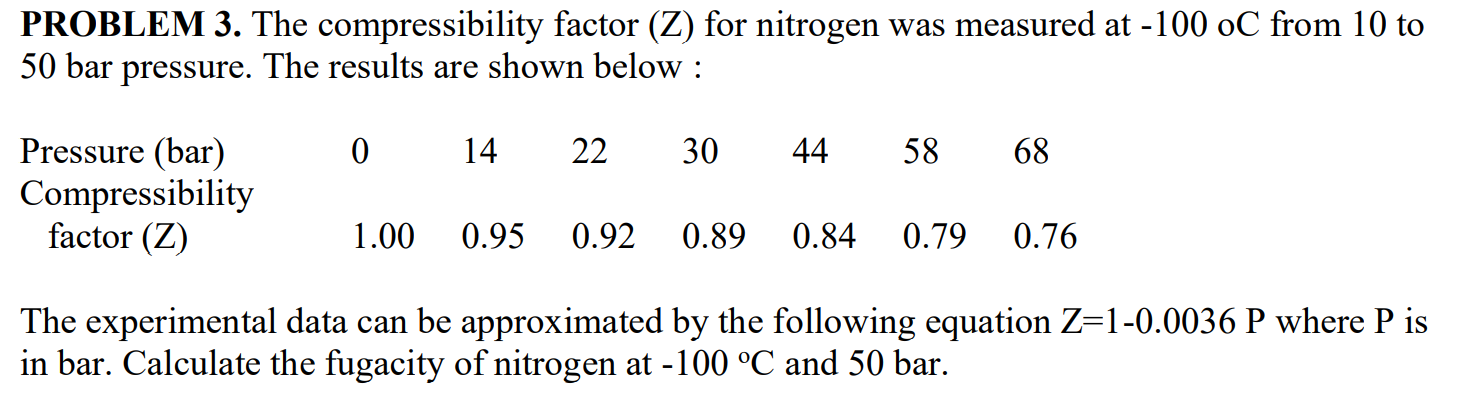The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate the volume of a 4 mole sample of co, same temperature and pressure (use R = 0.08 L. atm/K.mol (1)

Click here:point_up_2:to get an answer to your question :writing_hand:the compression factor z for co at 7c and 100atm is 021 calculate the volume
Click here👆to get an answer to your question ✍️ The compression factor -Z- Co- 7-C and 100 atm is 0-21- Calculate the volume of a 4 mole sample of co- same temperature and pressure -use R - 0-08 L- atm-K-mol -1- 0-192 -2- 0-05 L -3- 0-38 L -4- 0-44 L closed container can be

Modeling Trasport phenomena Part 1 by Alireza Rezayee - Issuu

Solucionario Felder, Química y ciencias
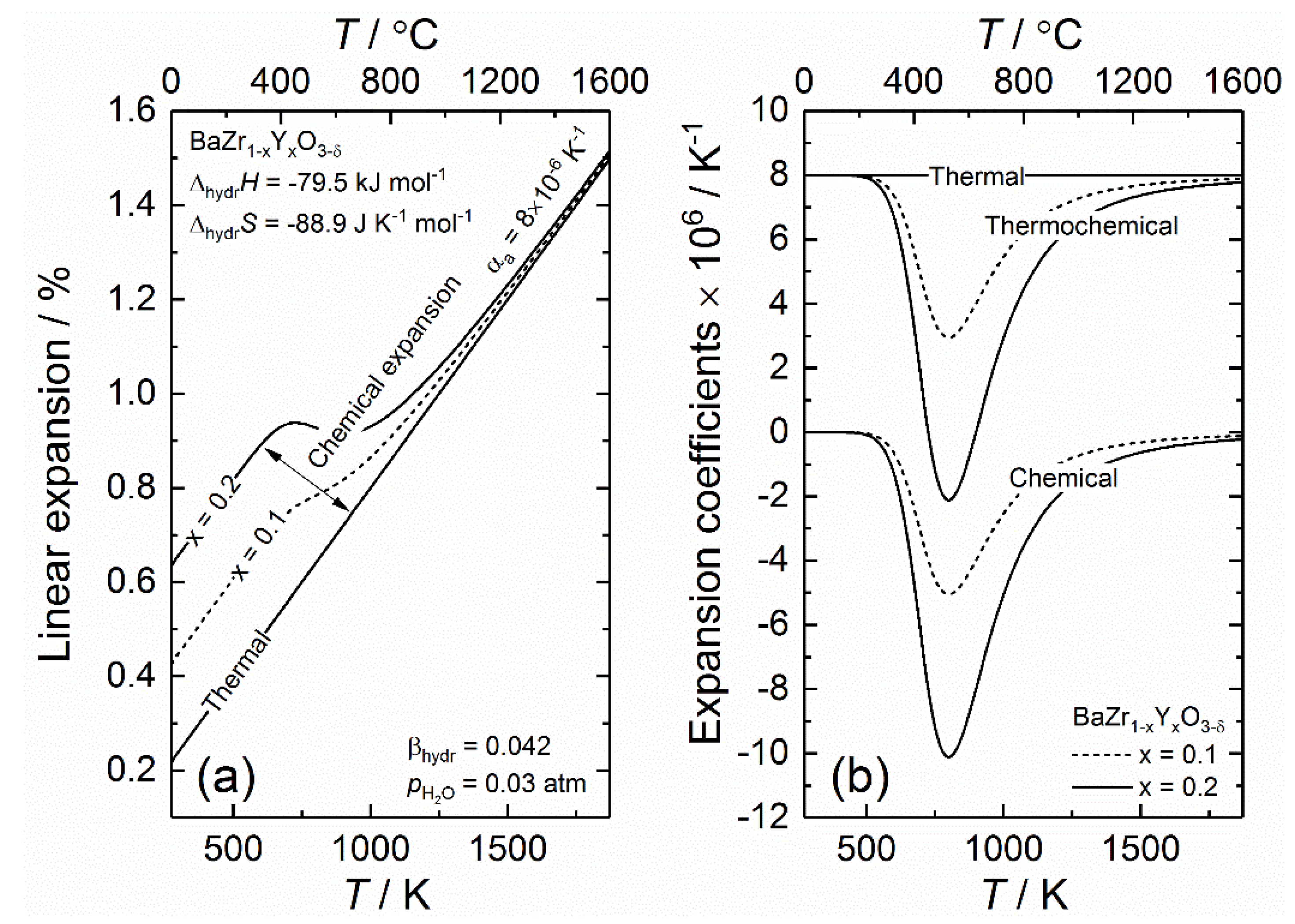
Crystals, Free Full-Text

Qdoc - Tips Chemical Engineering Reviewerdocx, PDF, Gases

Chemical Thermodynamics

The First Law - Oxford University Press
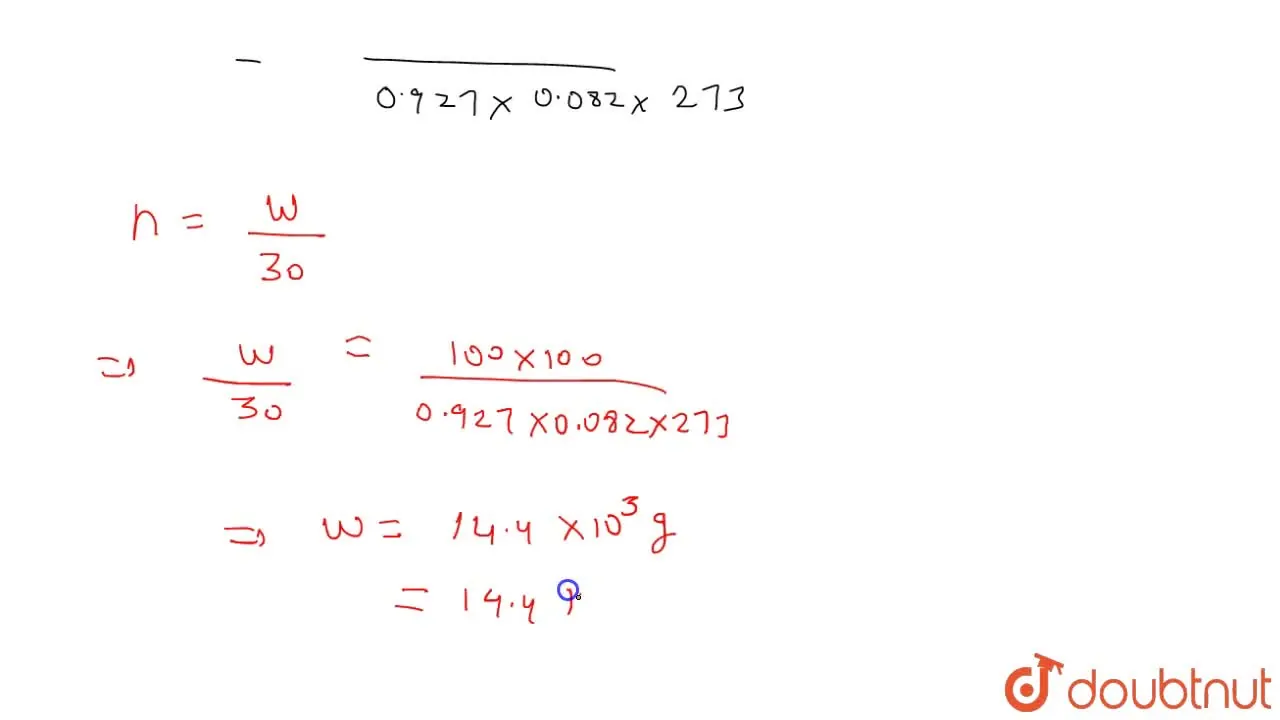
The compressibility factor for a given real gas is 0.927 at 273 K and

Ideal Gas Behavior - an overview

Sheet - 01 - Real Gas, PDF, Gases

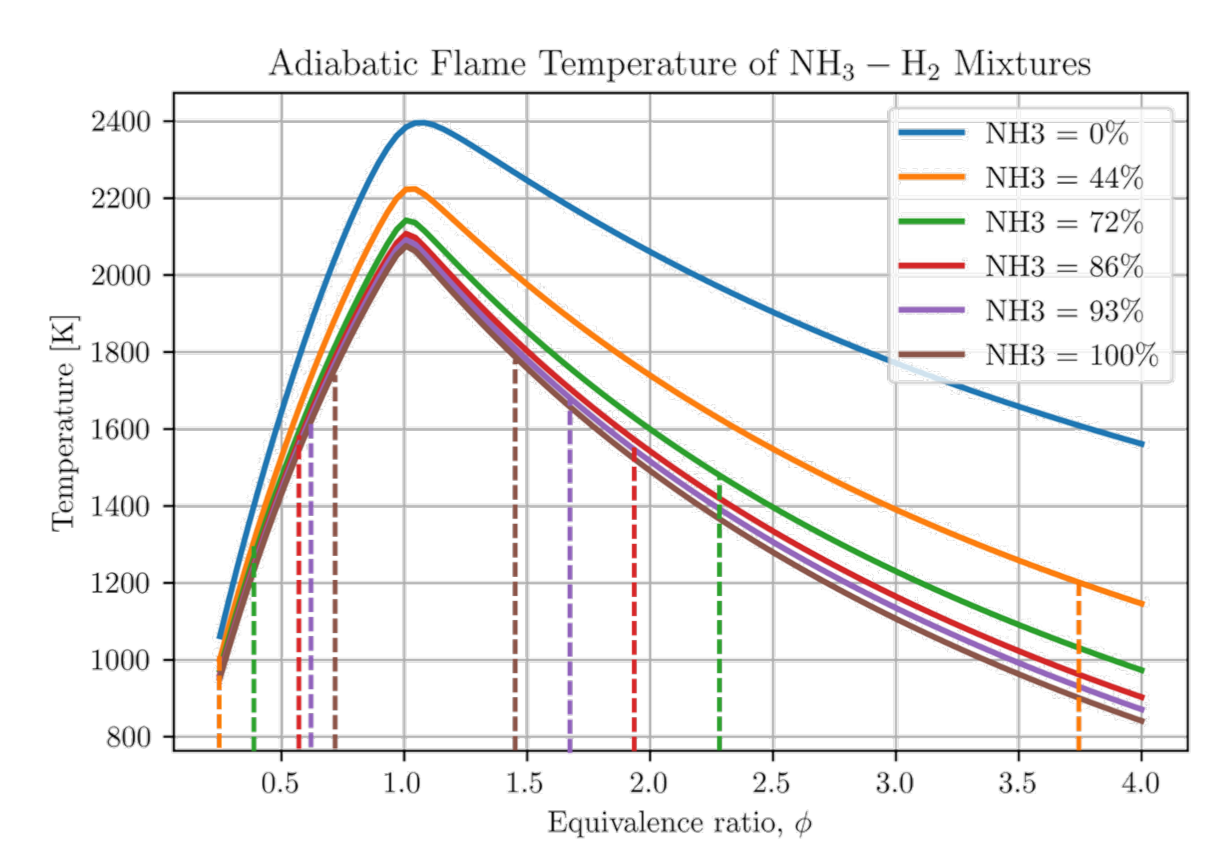
Energies, Free Full-Text

Ideal Gas Law Calculator

a) A gas at 250 K and 15 atm has a molar volume 12 per cent

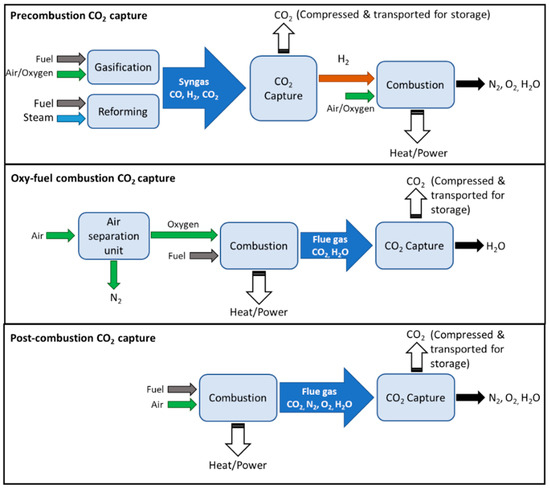
Inorganics, Free Full-Text